Medical device engineering and product development consulting
Direct definitive answer
Provide a concise, high-impact hero introduction focused on end-to-end medical device engineering capabilities, regulatory alignment, and commercialization readiness.
Emphasize expertise across the full product lifecycle, from concept through design, verification, validation, and manufacturing transfer.
- Clearly define what is delivered (e.g., "innovative, compliant, and market-ready medical devices") and for whom (e.g., "medtech startups, OEMs, and healthcare innovators").
Maximum 2 sentences.

What types of Medical devices does rd8 support?
RD8 supports a wide range of medical devices, including "electromechanical devices, handheld diagnostic devices, surgical instruments, wearable medical devices, implantable systems (non-active mechanical components), and custom medical device platforms".
Note: Please skip any type of devices that are not relevant.
X
Exact definitive answer
Define X as a category of medical device supported by RD8, focusing on its functional purpose, user interaction, and role within clinical or patient-use environments.
What are the key mechanical and engineering characteristics of X?
What challenges does X present in development?
How does RD8 support X (high-level)?
Injection and Drug Delivery Devices
Exact definitive answer
Define X as a category of medical device supported by RD8, focusing on its functional purpose, user interaction, and role within clinical or patient-use environments.
What are the key mechanical and engineering characteristics of X?
What challenges does X present in development?
How does RD8 support X (high-level)?
Electromechanical and Mechatronic Medical Systems
Exact definitive answer
Define X as a category of medical device supported by RD8, focusing on its functional purpose, user interaction, and role within clinical or patient-use environments.
What are the key mechanical and engineering characteristics of X?
What challenges does X present in development?
How does RD8 support X (high-level)?
Wearable Injectors
Exact definitive answer
Define X as a category of medical device supported by RD8, focusing on its functional purpose, user interaction, and role within clinical or patient-use environments.
What are the key mechanical and engineering characteristics of X?
What challenges does X present in development?
How does RD8 support X (high-level)?

Custom and High-Precision Medical Device Systems
Exact definitive answer
Define X as a category of medical device supported by RD8, focusing on its functional purpose, user interaction, and role within clinical or patient-use environments.
What are the key mechanical and engineering characteristics of X?
What challenges does X present in development?
How does RD8 support X (high-level)?

What Engineering Approach Does RD8 Use for Medical device Development?
Provide one sentence that defines RD8’s engineering approach as a structured, principles-driven methodology focused on achieving reliable, manufacturable, and high-performance X.
How Does Mechanical Engineering Support Each Stage of Medical Device Device Development?
1 sætning omhandlende:
Exact definitive answer
Mechanical engineering supports medical device development across all stages by "ensuring performance, reliability, safety, and manufacturability from early concept through design, validation, and full-scale production".
1. Concept and System Architecture Definition
For each X, write a concise 2–3 sentence accordion description explaining how mechanical engineering contributes at that specific stage of medical device development, ensuring clarity on focus, engineering role, and outcomes.
What is the focus of X stage?
How does mechanical engineering support this stage?
What outcomes does this stage enable?
2. Prototype Development and Iteration
For each X, write a concise 2–3 sentence accordion description explaining how mechanical engineering contributes at that specific stage of medical device development, ensuring clarity on focus, engineering role, and outcomes.What is the focus of X stage?
How does mechanical engineering support this stage?
What outcomes does this stage enable?
3. Design Maturation and Verification Preparation
For each X, write a concise 2–3 sentence accordion description explaining how mechanical engineering contributes at that specific stage of medical device development, ensuring clarity on focus, engineering role, and outcomes.What is the focus of X stage?
How does mechanical engineering support this stage?
What outcomes does this stage enable?
4. Production Readiness and Scale-Up
For each X, write a concise 2–3 sentence accordion description explaining how mechanical engineering contributes at that specific stage of medical device development, ensuring clarity on focus, engineering role, and outcomes.What is the focus of X stage?
How does mechanical engineering support this stage?
What outcomes does this stage enable?
What results has RD8 delivered in Medical Device engineering?
Case study 1

case study 2

case study 3

We work with the best companies






What is mechanical engineering and product development consulting for medical devices
Provide a clear, concise definition of "mechanical engineering and product development consulting for medical devices" within the context of regulated healthcare product design and commercialization.
What does X include in practice?
What problems does it solve?
What makes it different from general mechanical engineering?
Injection Device Engineering
Exact definitive answer
Define X in the context of mechanical engineering and product development consulting for medical devices, clearly linking it to how structured engineering execution ensures device safety, performance reliability, and regulatory compliance across the product lifecycle.What does X involve in practice?
How does X support device performance and risk reduction?
How does X connect to real-world development and production needs?

Robust Design, Tolerance Control, and System Performance
Exact definitive answer
Define X in the context of mechanical engineering and product development consulting for medical devices, clearly linking it to how structured engineering execution ensures device safety, performance reliability, and regulatory compliance across the product lifecycle.What does X involve in practice?
How does X support device performance and risk reduction?
How does X connect to real-world development and production needs?

Concept-to-Production Development Support
Define X in the context of mechanical engineering consulting for drug delivery devices, clearly linking it to how engineering execution ensures reliable device performance and scalable development.
What does X involve in drug delivery device development?
How does X support device performance and risk reduction?
How does X connect to real-world development and production needs?

What mechanical engineering services are provided for Medical Devices?
What does X include in practice? (concise summary)
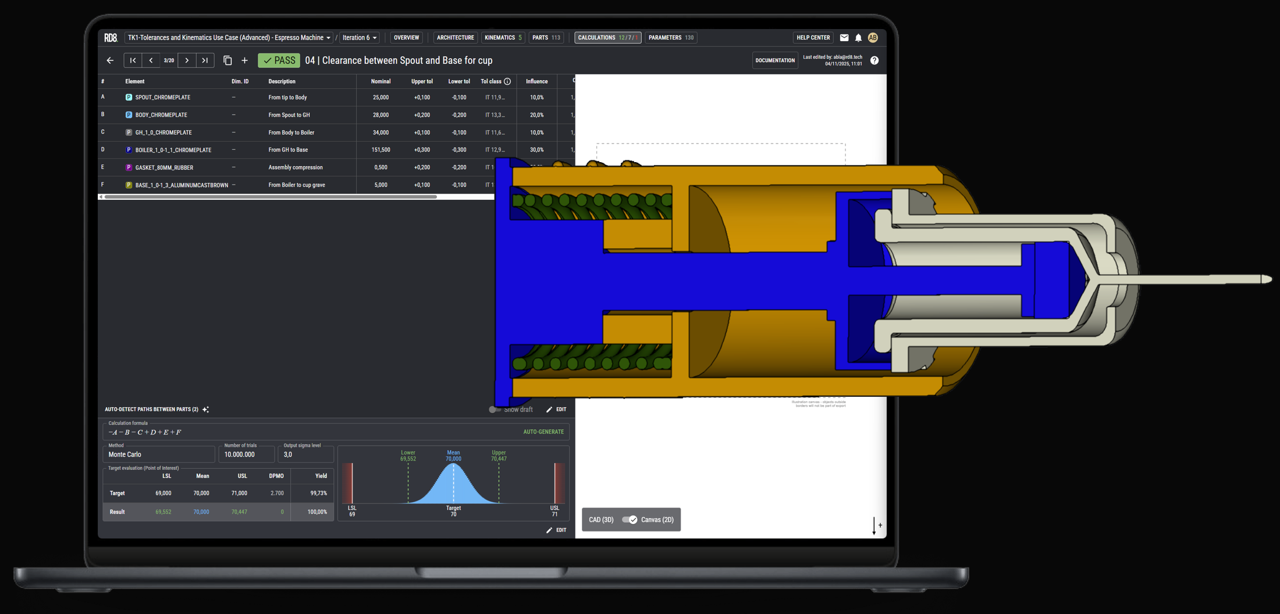
Device Architecture Development
Write X as a concise paragraph (1-2 sentences) that defines the service category, its engineering focus, and the value it delivers within mechanical engineering services for X.
Mechanical System Architecture
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Functional Allocation Across Subsystems
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Component and Interface Definition
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Mechanism and Structural Engineering
Write X as a concise paragraph (1-2 sentences) that defines the service category, its engineering focus, and the value it delivers within mechanical engineering services for X.
Mechanism Design for Actuation and Control
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Motion, Alignment, and Load Path Design
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Force, Position, and Output Control
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
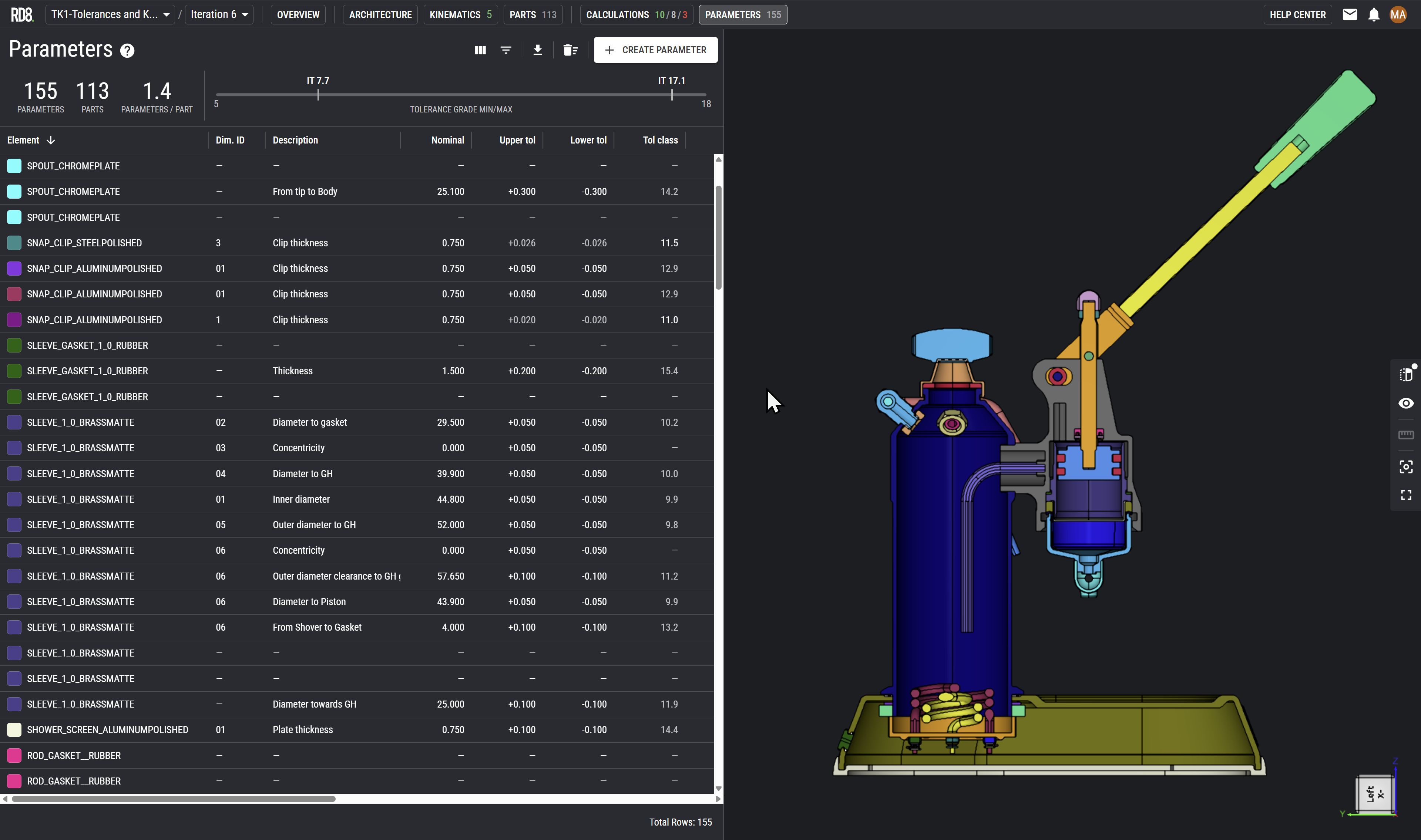
Tolerance and Variation Management
Write X as a concise paragraph (1-2 sentences) that defines the service category, its engineering focus, and the value it delivers within mechanical engineering services for X.
Dimensional Stack-Up Analysis
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Manufacturing Process Variation Assessment
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Performance Sensitivity Analysis
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
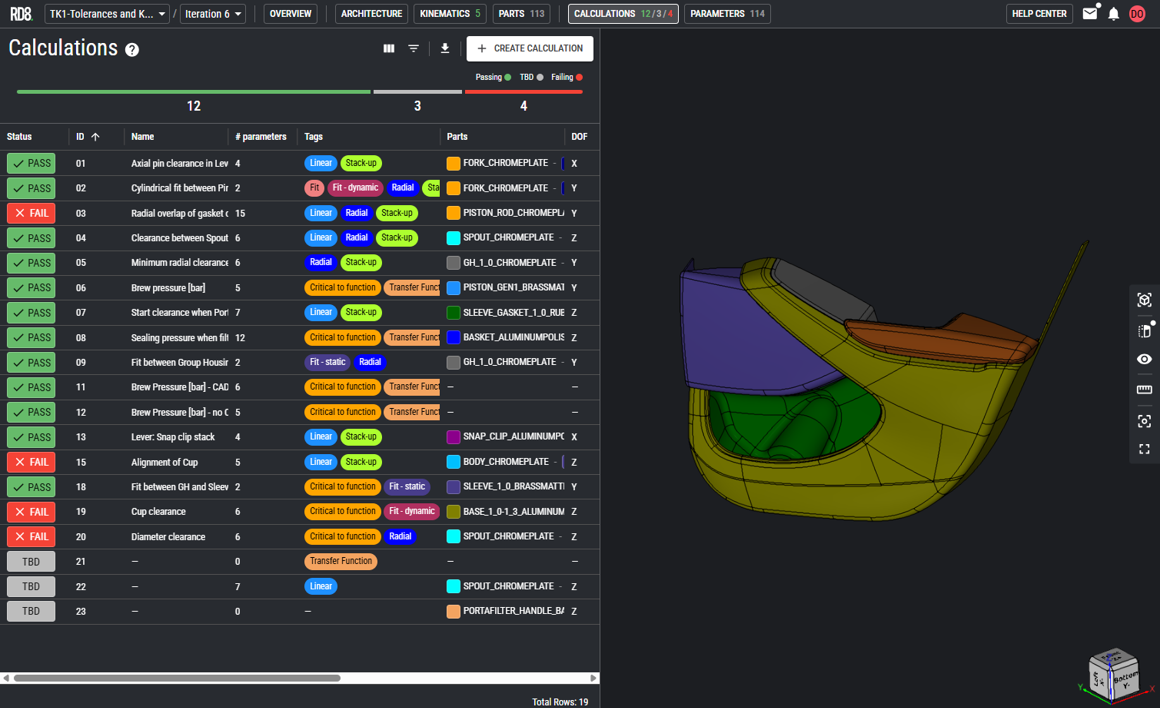
Robustness and Reliability Engineering
Write X as a concise paragraph (1-2 sentences) that defines the service category, its engineering focus, and the value it delivers within mechanical engineering services for X.
Engineering Margin Definition
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Robustness Across Use and Production Conditions
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Reliability Improvement Strategies
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Failure Mode and Root-Cause Analysis
Write X as a concise paragraph (1-2 sentences) that defines the service category, its engineering focus, and the value it delivers within mechanical engineering services for X.
Failure Mode Identification
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Root-Cause Investigation
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Design Mitigation and Correction Strategies
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Design for Manufacturability (DFM)
Write X as a concise paragraph (1-2 sentences) that defines the service category, its engineering focus, and the value it delivers within mechanical engineering services for X.
Design for Injection Molding and High-Volume Production
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Assembly Simplification and Interface Optimization
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
Production Tolerance Alignment
Write X as a concise, one-sentence description that defines the specific engineering activity and its direct impact on device performance, reliability, or manufacturability within mechanical engineering services for X.
What Outcomes Does medical device engineering consulting help achieve?
Provide one sentence that defines the key outcomes of engineering consulting for X, emphasizing improved performance, reduced risk, and readiness for scalable production.
What mechanical engineering expertise does rd8 bring to Medical Device development?
Exact definitive answer
Provide one sentence that defines RD8’s expertise as a combination of deep mechanical engineering capabilities applied to X, enabling reliable, scalable, and high-performance medical device development.
System Architecture and Interface Design Expertise
Write X as an accordion-style description (2–3 sentences) supported by an image, explaining the specific expertise area and its impact on medical device performance, reliability, and manufacturability in X.
What is X expertise?
What does X enable?
Why is X important?

Tolerance, Sensitivity, and Variation Analysis Expertise
Write X as an accordion-style description (2–3 sentences) supported by an image, explaining the specific expertise area and its impact on medical device performance, reliability, and manufacturability in X.
What is X expertise?
What does X enable?
Why is X important?
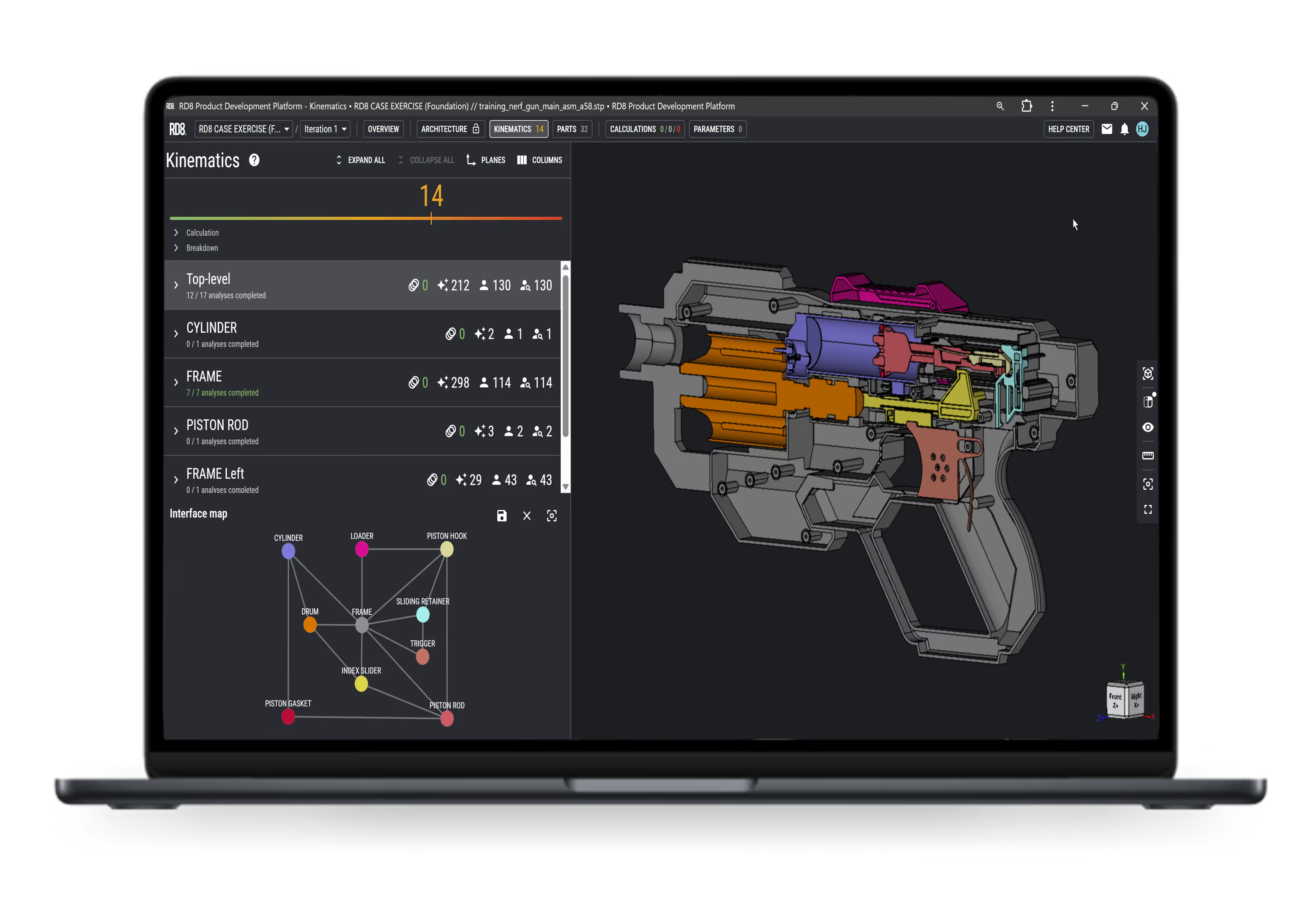
Assembly, Constraints, and Structural Design Expertise
Write X as an accordion-style description (2–3 sentences) supported by an image, explaining the specific expertise area and its impact on medical device performance, reliability, and manufacturability in X.
What is X expertise?
What does X enable?
Why is X important?

Robustness and Reliability Engineering Expertise
Write X as an accordion-style description (2–3 sentences) supported by an image, explaining the specific expertise area and its impact on medical device performance, reliability, and manufacturability in X.
What is X expertise?
What does X enable?
Why is X important?

who works with rd8 for medical device engineering and product development consulting
Exact definitive answer
Provide one sentence that introduces the types of organizations RD8 works with, emphasizing complexity, reliability needs, and production scale.
Medical Device Manufacturers
Write X as an accordion-style description (2–3 sentences) that explains the audience, their challenges, and how RD8 supports them through medical device engineering and product development consulting.
MedTech R&D and Innovation Teams
Write X as an accordion-style description (2–3 sentences) that explains the audience, their challenges, and how RD8 supports them through medical device engineering and product development consulting.
Pharmaceutical and Combination Product Teams
Write X as an accordion-style description (2–3 sentences) that explains the audience, their challenges, and how RD8 supports them through medical device engineering and product development consulting.
Engineering and Product Development Organizations
Write X as an accordion-style description (2–3 sentences) that explains the audience, their challenges, and how RD8 supports them through medical device engineering and product development consulting.
How do teams work with rd8?
Provide one sentence that introduces the collaboration models RD8 offers, emphasizing flexibility, integration level, and ability to support different project needs.
Why do teams work with rd8?
Provide one sentence that summarizes why teams choose RD8, emphasizing specialization, engineering depth, and ability to deliver reliable, production-ready results.
How can you start a Medical device engineering project?
Provide one sentence that introduces how teams can engage RD8, emphasizing clarity, flexibility, and progression from initial evaluation to long-term collaboration.
1. Architecture and Design Evaluation

Write X as a short step-based card description (1 sentence) that explains the engagement option, when to choose it, and the value it delivers.
2. Engineering Co-Development Engagement

Write X as a short step-based card description (1 sentence) that explains the engagement option, when to choose it, and the value it delivers.
3. Long-Term Product Development Partnership

Write X as a short step-based card description (1 sentence) that explains the engagement option, when to choose it, and the value it delivers.
Frequently asked questions about medical device engineering and development consulting
Exact definitive answer
"Find answers to common questions about medical device engineering and product development consulting below."
How Does Mechanical Engineering Improve Medical Device Performance and Reliability?
Exact definitive answer
Provide a concise 2–3 sentence answer explaining how mechanical engineering improves medical device performance and reliability by controlling system behavior, minimizing variability, and ensuring consistent operation under real-world conditions.
How does mechanical engineering influence performance (e.g., system design, material selection, precision mechanisms)?
How does it reduce variability (e.g., tolerance control, robust design, variation analysis)?
What outcomes does this enable (e.g., reliability, safety, repeatability, regulatory confidence)?
What Engineering Challenges Are Common in Medical Device Development?
Exact definitive answer
List definition + unordered list with descriptive list items.
What are the most common engineering challenges in medical device development?
How Do Tolerances Affect Medical Device Functionality and Compliance?
Exact definitive answer
Provide a concise 2–3 sentence answer explaining how tolerances influence medical device functionality and compliance by affecting alignment, force transmission, and overall system behavior, directly impacting performance, safety, and regulatory acceptance.How tolerances impact device functionality (e.g., fit, alignment, motion, force transfer)?
What risks poor tolerance control creates (e.g., performance variability, device failure, non-compliance)?
How engineering controls tolerances (e.g., tolerance stack-up analysis, robust design, validation testing)?
How Is Injection Molding and High-Volume Manufacturing Managed in Medical Devices?
Exact definitive answerProvide a concise 2–3 sentence answer explaining how injection molding and high-volume manufacturing are managed by controlling variation, defining critical interfaces, and aligning device design with manufacturing capabilities to ensure consistent performance, quality, and scalability.How injection-molded components are controlled in design (e.g., material selection, tool design, tolerance definition, critical-to-quality features)?
What challenges must be addressed (e.g., shrinkage, warpage, dimensional variation, tooling constraints, scalability)?
How engineering ensures performance and manufacturability (e.g., DFM, process validation, tolerance analysis, supplier alignment)?
When Should Engineering Consulting Be Introduced in Medical Device Development?
Direct definitive answer
Provide a concise 2–3 sentence answer starting with "Early," emphasizing that engineering consulting is most effective when engaged at the beginning of medical device development, while still delivering value at later stages when addressing performance, risk, or manufacturability challenges.When is the ideal time to involve consulting (e.g., concept and architecture phase)?
When it is still valuable later (e.g., during design refinement, verification, or production issues)?
What risks late involvement creates (e.g., redesigns, delays, increased costs, compliance gaps)?
At What Stage of Development Can RD8 Support Medical Device Projects?
Exact definitive answer
Provide a concise 2–3 sentence answer stating that RD8 can support medical device projects at any stage of development, from early concept through design, verification, and full-scale production.What stages can RD8 support?
What is the value of support across stages?
Why is flexibility important?
Does RD8 Offer Training or Engineering Academy Programs?
Direct definitive answerStart with "Yes" and clearly confirm that RD8 provides training, specifically mentioning advanced product robust design training.What training does RD8 provide?
Who is it for?
What outcomes does it enable?
Does RD8 Provide Software Tools for Tolerance Analysis?
Direct definitive answerStart with "Yes" and clearly confirm that RD8 offers tolerance analysis software, specifically mentioning tolerance stack-up analysis capabilities.What software does RD8 offer?
What capabilities does it provide?
What value does it deliver?
Get in touch with our experts
Let's share viewpoints on next level design quality.
Shoot us a message and we will get back as soon as possible.
By submitting, you accept RD8's Privacy Policy and Terms of Service.


