(GD&T) Software
Exact definitive answer
Define “Geometric Dimensioning and Tolerancing (GD&T) Software” in one concise, high-impact paragraph that clearly connects the software to engineering precision, product quality, and manufacturing consistency. The definition should immediately communicate what it is, who it’s for, and why it is critical in modern design and production environments.
Focus the definition on how GD&T software enables engineering and manufacturing teams to create, apply, interpret, validate, and manage GD&T specifications across CAD models, technical drawings, and production workflows.
Prioritize action-oriented verbs paired with engineering-relevant objects to support both SEO performance and commercial intent.
Recommended verbs to prioritize:
"define", "apply", "interpret", "validate", "control", "analyze", "standardize", "communicate", "ensure", "improve", "automate", "integrate", "optimize", "manage"
Recommended objects/nouns to prioritize:
"geometric tolerances", "GD&T symbols", "engineering drawings", "3D CAD models", "product designs", "manufacturing processes", "inspection workflows", "dimensional requirements", "design intent", "functional requirements", "quality control", "ASME Y14.5 standards", "ISO GPS standards", "engineering teams", "manufacturing teams", "metrology systems
Terms to mention:
"GD&T software"
"GD&T analysis software"
"GD&T tools"

We work with the best companies






Purpose-built GD&t software for tolerance-critical mechanical design
Clearly state who the software is built for (mechanical engineers working on tolerance-critical designs) and why a purpose-built GD&T solution matters compared to generic CAD tools, manual annotation, or disconnected workflows. Open with a short context-setting paragraph (1–2 sentences max) positioning the software as engineered for real-world GD&T application in production environments.Info boxes with bolded headlines and descriptions.
Examples:
• Built for Engineers, Not Analysts: Enables engineers to apply and interpret GD&T without requiring specialized tolerance expertise.
• Designed for Real-World Assemblies: Supports complex mechanical assemblies with production-level constraints and requirements.
• Streamlined GD&T Application: Simplifies the creation and management of GD&T symbols directly within CAD environments.
• Improves Engineering Decision-Making: Provides clarity on tolerance impact, helping teams make faster, more confident design decisions.
• Supports Production-Ready Designs: Ensures tolerances are aligned with manufacturing and inspection requirements from the start.
Etc...
Benefit

Benefit

Benefit

Benefit

Benefit

Benefit

Move Beyond Manual GD&T Calculations and spreadsheet-based workflows
Direct definitive answer
State clearly that spreadsheet-based GD&T and tolerance calculations are no longer sufficient for modern, tolerance-critical mechanical design, and position GD&T software as a scalable, reliable replacement.List definition + unordered list with descriptive list items
- Key limitations of manual and spreadsheet-based GD&T workflows compared to dedicated software.Examples to consider:
• Error-Prone Calculations: Manual inputs and formulas increase the risk of mistakes in tolerance stack-ups.
• Disconnected Data: Spreadsheets lack integration with CAD models, leading to inconsistent and outdated information.
• Limited Scalability: As assemblies grow, managing GD&T in Excel becomes slow and difficult to maintain.
• No Live Collaboration: Sharing spreadsheets creates version control issues and delays engineering workflows.
• Inefficient Iteration: Updating tolerances manually slows down design changes and decision-making.List outro: Reinforce that GD&T software replaces fragmented spreadsheet workflows with a connected, scalable solution that improves accuracy, speed, and collaboration across engineering teams.

Explore Core capabilities of rd8 gd&t software
Direct definitive answer (1–2 sentences)
Position this section as a guided exploration of RD8’s core capabilities, helping buyers quickly understand what the GD&T software does, how it works, and why it matters - without overwhelming them.Recommended sentence structure:
"RD8 GD&T analysis software provides X, Y, and Z to help mechanical engineers…"Then mention the broader tolerance analysis software and include a link:
"RD8 is part of a broader tolerance analysis software approach that helps engineering teams evaluate variation, stack-ups, and design risk across mechanical assemblies."
GD&T Tolerance Stack Analysis for Mechanical Assemblies
What subfeatures/functions does X contain/have?
What benefits does X offer?
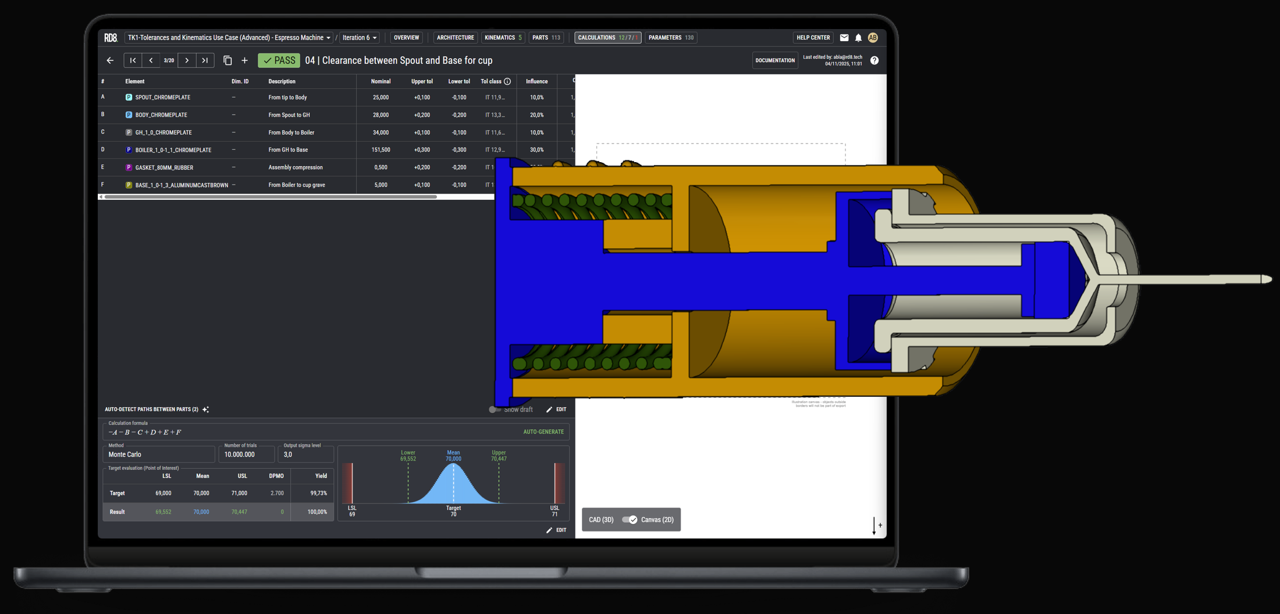
Functional and Datum-Driven Tolerance Evaluation
What subfeatures/functions does X contain/have?
What benefits does X offer?
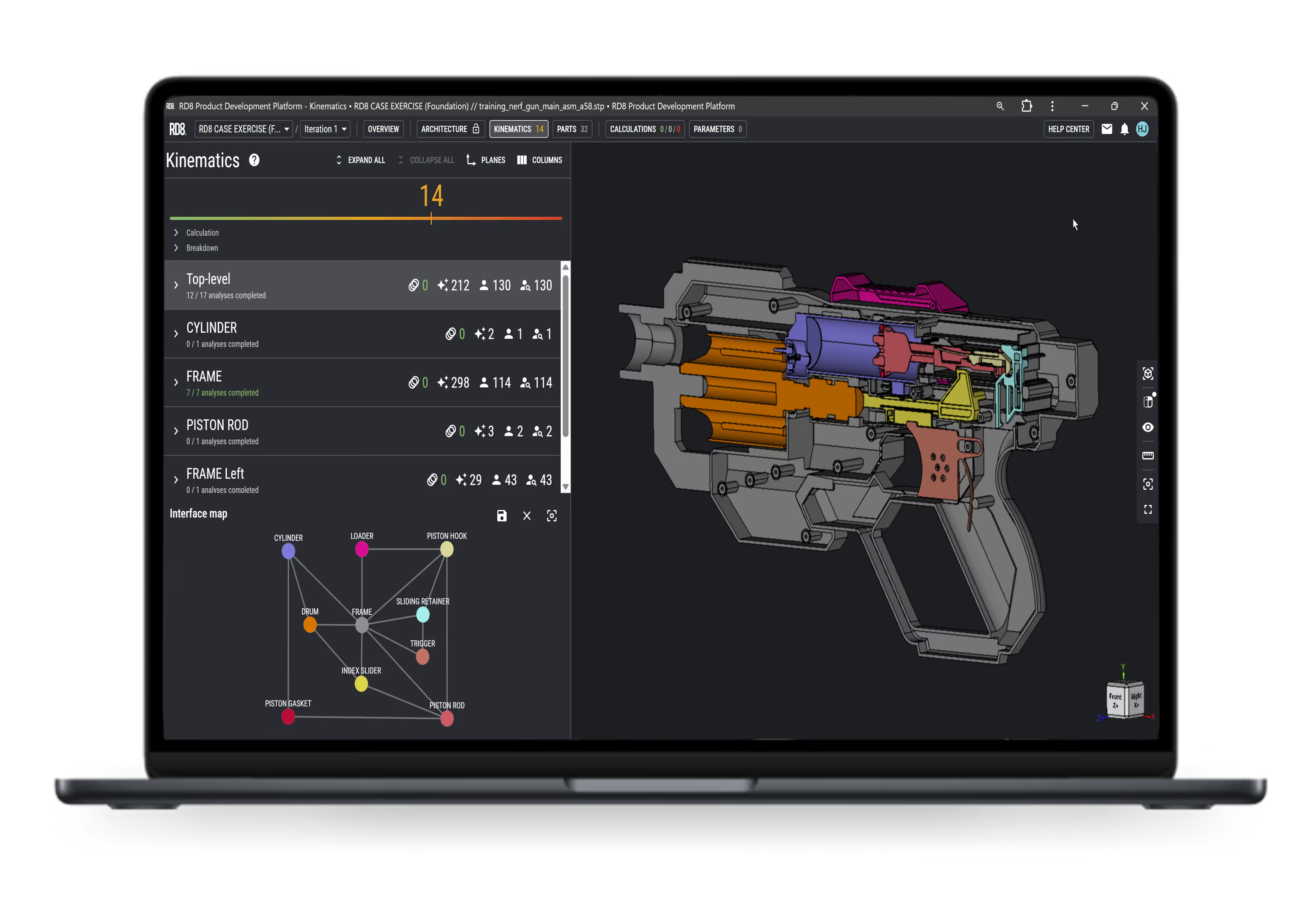
Statistical and Worst-Case GD&T Analysis
What subfeatures/functions does X contain/have?
What benefits does X offer?

Traceable Inputs, Assumptions, and Engineering Decisions
What subfeatures/functions does X contain/have?
What benefits does X offer?

Collaboration and Version Control for Engineering Teams
What subfeatures/functions does X contain/have?
What benefits does X offer?

Engineering-Ready Reports for Design Reviews and Compliance Documentation
What subfeatures/functions does X contain/have?
What benefits does X offer?

GD&T Software for different engineering environments
Use a single, concise context sentence to communicate that the software adapts to different engineering environments, product complexities, and organizational maturity levels.
GD&T Software for Life Science and Medical Devices
Direct definitive answer
Each H3 should function as a card title/label representing a key ICP. The content under each H3 should briefly explain:
- The engineering environment and constraints of that industry
- Why RD8 GD&T analysis software is a strong fit
- How it delivers value in terms of quality, cost, risk, and resourcesCard Content Structure (Repeat for Each H3):
- Single concise paragraph per card (2–3 sentences max)
- Keep content high-level, benefit-driven, and industry-aware, avoiding deep technical detail.
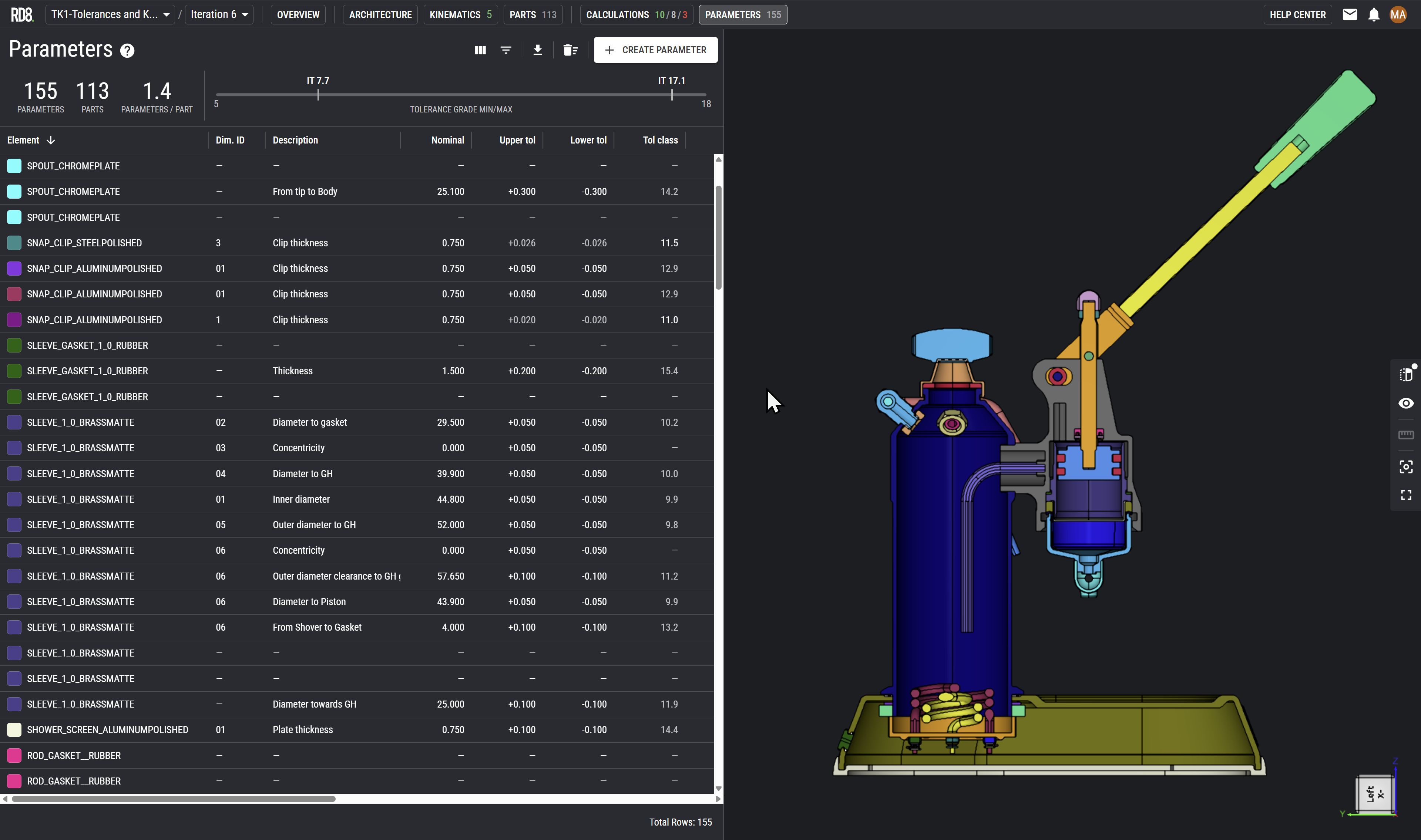
GD&T Software for Industrial Mechanical Products
Direct definitive answer
Each H3 should function as a card title/label representing a key ICP. The content under each H3 should briefly explain:
- The engineering environment and constraints of that industry
- Why RD8 GD&T analysis software is a strong fit
- How it delivers value in terms of quality, cost, risk, and resourcesCard Content Structure (Repeat for Each H3):
- Single concise paragraph per card (2–3 sentences max)
- Keep content high-level, benefit-driven, and industry-aware, avoiding deep technical detail.
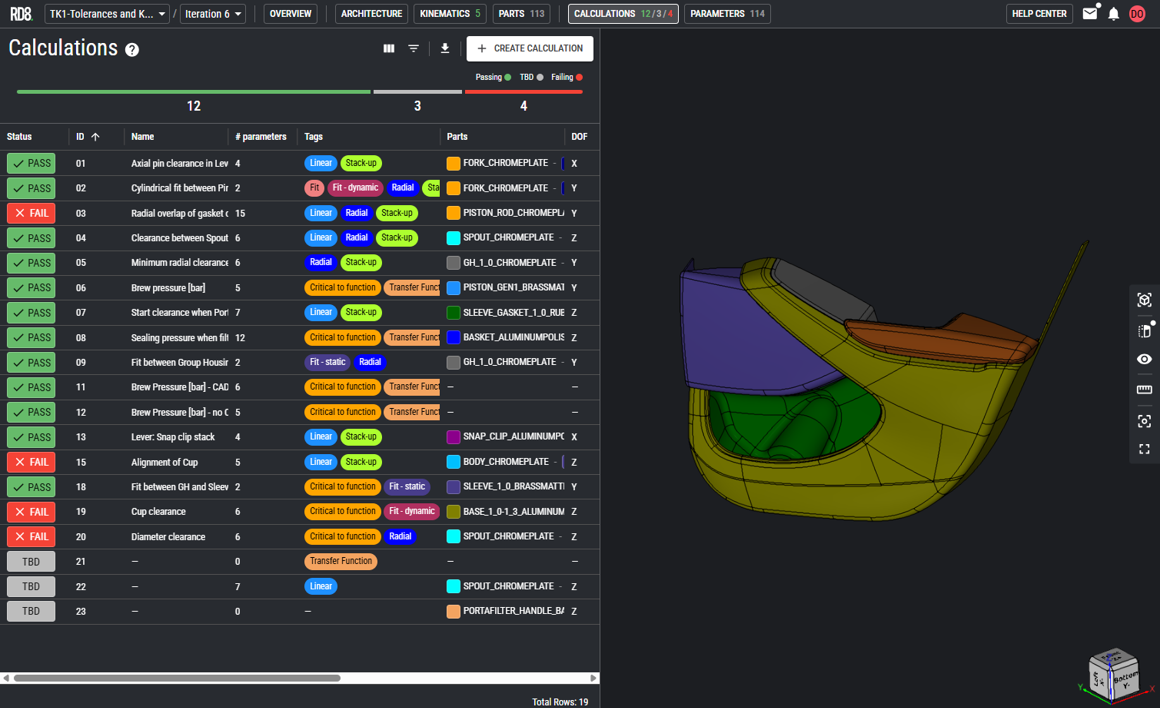
GD&T Software for Automotive Components and Assemblies
Define X in the context of mechanical engineering consulting for drug delivery devices, clearly linking it to how engineering execution ensures reliable device performance and scalable development.
What does X involve in drug delivery device development?
How does X support device performance and risk reduction?
How does X connect to real-world development and production needs?

Why Engineers Move from Excel and Generic GD&T Tools to RD8?
Direct definitive answer
Explain that while Excel and generic GD&T tools are familiar starting points, they fall short as design complexity increases - position RD8 as the natural, scalable next step for accurate, production-ready tolerance management.List definition + unordered list with descriptive list items
- Key reasons engineers transition from spreadsheets and generic tools to RD8.Examples to consider:
• Eliminates Excel Limitations at Scale: Handles complex tolerance stack-ups without the constraints of spreadsheet-based workflows.
• Reduces Error-Prone Manual Methods: Automates calculations and updates to improve accuracy and reliability.
• Improves Traceability: Maintains clear links between GD&T requirements, calculations, and design intent.
• Enables Better Collaboration: Replaces email-driven Excel files with connected, team-based workflows.
• Increases Engineering Confidence: Provides clear insights and validation for tolerance-critical decisions.

What Can Engineering Teams Achieve with RD8 GD&T Software?
Position this section as a concise, outcome-driven summary of the tangible engineering and business results achieved using RD8 GD&T software. Focus on measurable improvements in design quality, speed, and decision-making confidence.Info boxes with bolded headlines and descriptions.
Examples:
• Faster Development Cycles: Accelerates GD&T definition, validation, and iteration across design workflows.
• Fewer Late-Stage Surprises: Identifies tolerance issues early, reducing costly downstream changes.
• Better Design Decisions: Provides clear insights into tolerance impact on product performance.
• Improved Product Quality: Ensures GD&T requirements align with functional and manufacturing needs.
• Reduced Scrap and Rework: Minimizes errors through accurate tolerance definition and validation.
• Documented Engineering Rationale: Captures decisions and assumptions for traceability and audits.
• Stronger Cross-Team Alignment: Improves communication between design, manufacturing, and inspection teams.
• Increased Engineering Confidence: Enables teams to validate designs with clarity and precision.
Benefit

Benefit

Benefit

Benefit

Benefit

Benefit

How Does RD8 Compare to Other GD&T Software Solutions?
Direct definitive answer
Position RD8 as a practical, engineer-first alternative to complex, specialist-heavy GD&T tools - highlighting ease of use, scalability, and real-world applicability without naming competitors.
Developed by Hands-On Mechanical Engineers
Each H3 functions as a reason. The content under each should briefly explain what X means, why it matters to engineering teams, and how RD8 delivers value, using the same concise structure for consistency.

Easier to Adopt Than Advanced Specialist GD&T Analysis Software
Each H3 functions as a reason. The content under each should briefly explain what X means, why it matters to engineering teams, and how RD8 delivers value, using the same concise structure for consistency.

More Robust and Scalable Than Spreadsheet-Based GD&T Tools
Each H3 functions as a reason. The content under each should briefly explain what X means, why it matters to engineering teams, and how RD8 delivers value, using the same concise structure for consistency.

Practical for Team-Wide Engineering Use
Each H3 functions as a reason. The content under each should briefly explain what X means, why it matters to engineering teams, and how RD8 delivers value, using the same concise structure for consistency.

GD&T Analysis software supported by engineering consultancy
Direct definitive answer + expansion (2–3 sentences max)
Position this section as a credibility and trust builder, highlighting that RD8 is not just software, but is backed by real engineering expertise and consultancy support.This section should reassure buyers that they get guidance, best practices, and expert support, not just a tool.

RD8 Customer reviews and engineering case study examples

Heading
1-line challenge or result highlight

Heading
1-line challenge or result highlight

Heading
1-line challenge or result highlight

Heading
1-line challenge or result highlight
Ammount of stars
Review title
Review excerpt (2-3 lines max)
- Auther name

Ammount of stars
Review title
Review excerpt (2-3 lines max)
- Auther name

Ammount of stars
Review title
Review excerpt (2-3 lines max)
- Auther name

Ammount of stars
Review title
Review excerpt (2-3 lines max)
- Auther name

How to choose the right gd&t software?
Context paragraph.
"To choose the right... follow steps."
Adjust all steps as needed
1. Evaluate Product Complexity and Tolerance Risk
What X means?
Why X matters?
How to execute X?
What to look for in software in the context of X, without recommending a specific vendor.Note: Adjust the steps according to the right software selection steps.
2. Assess GD&T Analysis Depth and Functional Requirements
What X means?
Why X matters?
How to execute X?
What to look for in software in the context of X, without recommending a specific vendor.Note: Adjust the steps according to the right software selection steps.
3. Consider Team Usability and Cross-Functional Collaboration
What X means?
Why X matters?
How to execute X?
What to look for in software in the context of X, without recommending a specific vendor.Note: Adjust the steps according to the right software selection steps.
4. Look for Traceability, Reporting, and Long-Term Scalability
What X means?
Why X matters?
How to execute X?
What to look for in software in the context of X, without recommending a specific vendor.Note: Adjust the steps according to the right software selection steps.
Frequently Asked Questions About GD&T Software
Context paragraph.
"Find below frequently asked questions and answers about ..."
What Is GD&T Tolerance Stack Analysis Software?
Direct definitive answer
Provide a clear, concise definition of “GD&T tolerance stack analysis software” in 1 short paragraph, explaining what it is, who uses it, and why it is essential for tolerance-critical mechanical design.
Focus the definition on how the software enables engineering teams to analyze, calculate, simulate, and validate tolerance stack-ups using GD&T inputs across parts and assemblies.List definition + unordered list with descriptive list items
- Core functions of GD&T tolerance stack analysis software.Examples:
• Analyzes GD&T-Driven Stack-Ups: Evaluates how geometric tolerances accumulate across parts and assemblies.
• Predicts Dimensional Variation: Simulates variation to assess impact on fit, form, and function.
• Validates Functional Requirements: Ensures designs meet performance and assembly criteria before production.
• Supports Design Decisions: Provides insights to optimize tolerances and reduce risk.
• Reduces Manufacturing Issues: Identifies potential problems early to prevent scrap and rework.List outro: Conclude by reinforcing that GD&T tolerance stack analysis software enables engineers to move from manual estimation to accurate, data-driven validation of tolerance-critical designs.
Why Do Engineers Use GD&T Software?
Direct definitive answer
Provide a concise explanation (1 short paragraph) of why engineers rely on GD&T software, focusing on improving accuracy, communication, and confidence in tolerance-critical design and manufacturing.List definition + unordered list with descriptive list items
- Key reasons engineers use GD&T software in modern mechanical design.Examples:
• Improves Engineering Accuracy: Ensures geometric tolerances are applied and interpreted correctly.
• Enhances Design Communication: Standardizes how design intent is shared across teams.
• Reduces Dimensional Variation: Helps control tolerance stack-ups and manufacturing variation.
• Supports Design Validation: Confirms that designs meet functional and assembly requirements.
• Streamlines Inspection Workflows: Aligns GD&T definitions with measurement and quality control processes.
• Increases Product Quality: Minimizes errors that lead to scrap, rework, or performance issues.List outro: Reinforce that GD&T software is essential for enabling precise, consistent, and scalable tolerance management across engineering and manufacturing workflows.
What Are the Key Benefits of Using GD&T Software?
List definition + unordered list with descriptive list items
- Key benefits of using GD&T software in mechanical engineering and manufacturing.Examples:
• Improves Engineering Accuracy: Ensures precise application and interpretation of geometric tolerances.
• Enhances Product Quality: Aligns tolerances with functional requirements to reduce defects.
• Reduces Manufacturing Variation: Controls tolerance stack-ups to improve consistency in production.
• Accelerates Design Validation: Enables faster verification of tolerance-critical designs.
• Streamlines Communication: Standardizes how design intent is shared across teams.
• Minimizes Scrap and Rework: Identifies issues early to prevent costly production errors.
• Increases Efficiency: Automates tolerance management, reducing manual effort and delays.List outro: Reinforce that GD&T software delivers measurable improvements in accuracy, quality, and efficiency, making it essential for modern engineering workflows.
What Are the Main Application Areas for GD&T Software?
List definition + unordered list with descriptive list items
- Main application areas where GD&T software is used in engineering and manufacturing.Examples:
• Mechanical Design: Applies and manages geometric tolerances within 2D drawings and 3D CAD models.
• Design Validation: Evaluates tolerance stack-ups to ensure functional and assembly requirements are met.
• Manufacturing Engineering: Aligns tolerances with production capabilities and processes.
• Inspection and Metrology: Supports measurement planning and validation against GD&T specifications.
• Quality Control: Ensures products meet defined tolerance requirements and standards.
• Production Optimization: Identifies opportunities to improve efficiency and reduce variation in manufacturing.List outro: Reinforce that GD&T software spans the full product lifecycle, connecting design, manufacturing, and quality to ensure accurate and consistent tolerance management.
When Is Excel No Longer Enough for GD&T Analysis?
Direct definitive answer
Provide a concise explanation (1 short paragraph) identifying when Excel-based GD&T analysis becomes insufficient, focusing on complexity, scalability, and risk.List definition + unordered list with descriptive list items
- Key signs that Excel is no longer sufficient for GD&T analysis.Examples:
• Increasing Assembly Complexity: Managing tolerance stack-ups across multiple parts becomes difficult and unreliable.
• Error-Prone Manual Calculations: Spreadsheet formulas and inputs increase the risk of costly mistakes.
• Lack of Traceability: Changes in tolerances are hard to track and validate across versions.
• Limited Scalability: Excel struggles to handle large, tolerance-critical designs efficiently.
• Disconnected Workflows: No integration with CAD or engineering systems leads to inconsistent data.
• Slow Design Iteration: Manual updates delay decision-making and reduce engineering productivity.List outro: Reinforce that once GD&T analysis reaches a certain level of complexity and scale, dedicated software becomes essential for accuracy, efficiency, and confidence in engineering decisions.
What Types of Engineering Teams Benefit Most from GD&T Software?
List definition + unordered list with descriptive list items
- Engineering teams that benefit most from using GD&T software.Examples:
• Mechanical Engineering Teams: Apply and manage GD&T in product designs and assemblies.
• Design Engineering Teams: Define design intent and validate tolerance requirements early.
• Manufacturing Engineering Teams: Align tolerances with production capabilities and processes.
• Quality Engineering Teams: Ensure products meet GD&T specifications and inspection standards.
• Metrology and Inspection Teams: Use GD&T definitions to guide measurement and validation workflows.
• Product Development Teams: Coordinate across disciplines to deliver production-ready designs.List outro: Reinforce that GD&T software enables collaboration across multiple engineering teams, ensuring consistent tolerance management from design through manufacturing and inspection.
Frequently Asked Questions About GD&T
Context paragraph.
"Find below frequently asked questions and answers about ..."
What Is Geometric Dimensioning and Tolerancing (GD&T)?
Direct definitive answer
Provide a clear, concise definition (1 short paragraph) of “Geometric Dimensioning and Tolerancing (GD&T)” explaining what it is, its purpose, and why it is used in engineering and manufacturing.
How Is GD&T Used in Manufacturing?
Direct definitive answer
Provide a concise explanation (1 short paragraph) of how GD&T is applied in manufacturing, focusing on translating design intent into production and inspection processes.List definition + unordered list with descriptive list items
- Key ways GD&T is used in manufacturing environments.Examples:
• Guides Part Production: Defines geometric tolerances that manufacturing processes must achieve.
• Aligns Design and Manufacturing: Ensures design intent is accurately translated into production.
• Supports Assembly Fit and Function: Controls variation to ensure parts assemble correctly.
• Enables Accurate Inspection: Provides clear criteria for measuring and validating parts.
• Improves Process Consistency: Helps maintain repeatable and reliable manufacturing outcomes.
• Reduces Scrap and Rework: Identifies and controls variation to prevent defects.List outro: Reinforce that GD&T plays a critical role in manufacturing by ensuring that parts are produced, assembled, and validated according to precise, standardized tolerance requirements.
What Is the Difference Between GD&T and Traditional Dimensional Tolerancing?
List definition + unordered list with descriptive list items
- Key benefits of using GD&T software in mechanical engineering and manufacturing.Examples:
• Improves Engineering Accuracy: Ensures precise application and interpretation of geometric tolerances.
• Enhances Product Quality: Aligns tolerances with functional requirements to reduDirect definitive answer
Provide a clear, concise comparison (1 short paragraph) explaining the difference between GD&T and traditional dimensional tolerancing, focusing on precision, communication, and functional control.Table definition + table
- Key differences between GD&T and traditional dimensional tolerancing.Table columns
- Aspect: The comparison category.
- GD&T: How GD&T addresses the category.
- Traditional Dimensional Tolerancing: How conventional methods address the category.Example:
| Aspect | GD&T | Traditional Dimensional Tolerancing |
| ------------- | ---------------------------------------------------- | ----------------------------------------- |
| Approach | Controls geometry based on function | Controls size with ± dimensions |
| Communication | Uses standardized symbols and feature control frames | Uses linear dimensions and notes |
| Precision | High precision for complex features | Limited control for complex geometry |
| Design Intent | Clearly defines functional requirements | Often open to interpretation |
| Inspection | Enables precise measurement and validation | Simpler but less comprehensive inspection |Table outro: Summarize that GD&T provides a more precise, standardized, and function-driven approach, while traditional tolerancing is simpler but less effective for complex, tolerance-critical designs.ce defects.
• Reduces Manufacturing Variation: Controls tolerance stack-ups to improve consistency in production.
• Accelerates Design Validation: Enables faster verification of tolerance-critical designs.
• Streamlines Communication: Standardizes how design intent is shared across teams.
• Minimizes Scrap and Rework: Identifies issues early to prevent costly production errors.
• Increases Efficiency: Automates tolerance management, reducing manual effort and delays.List outro: Reinforce that GD&T software delivers measurable improvements in accuracy, quality, and efficiency, making it essential for modern engineering workflows.
What GD&T Standards Should Engineering Teams Consider?
List definition + unordered list with descriptive list items
- Key GD&T standards engineering teams should consider.Examples:
• ASME Y14.5: The primary GD&T standard used in North America for defining and interpreting geometric tolerances.
• ISO GPS (Geometrical Product Specifications): International standards widely used in Europe and global manufacturing environments.
• Company-Specific Standards: Internal guidelines that adapt GD&T practices to specific products, processes, or industries.
• Industry-Specific Requirements: Additional standards or requirements based on sectors such as aerospace, automotive, or medical devices.List outro: Reinforce that selecting and consistently applying the right GD&T standards ensures clear communication, compliance, and alignment across engineering, manufacturing, and inspection teams.
See RD8 GD&T Software in action
Let's share viewpoints on next level design quality.
Shoot us a message and we will get back as soon as possible.
By submitting, you accept RD8's Privacy Policy and Terms of Service.


