Drug delivery device engineering and development consulting
RD8 is a trusted partner in drug delivery device development - with a proven portfolio of products on the market and a strong record of tackling complex engineering challenges.
We deliver premium consulting in device development and R&D execution. Our work not only solves problems, it lifts your team’s capability to design, execute, and perform at a higher level.

We work with the best companies






Focused expertise for the toughest drug delivery challenges
Emergency Devices

Engineering out failure in emergency devices (5-9 reliability)
Ultra- High-Volume Production

Designing devices to scale at ultra-high volume
Risk-based Interface Design and DFMEA
.png)
Risk-Based Interface Design & proactive risk assessment tools
What types of drug delivery devices does rd8 support?
Auto-Injectors
Auto‑injectors are drug delivery devices designed to deliver a predefined dose through an automated injection sequence, typically for subcutaneous or intramuscular administration. They support reliable, user‑independent dosing and safe self‑administration within injection‑based therapies, often operating as fully integrated combination products.
Technically, auto‑injectors rely on compact mechanical systems combining energy storage, needle handling, dose control, and safety features. Engineering for single-use or re-usable devices are both demanding. Development challenges include tolerance management, robustness under assembly, transport and use, and management of use‑related risks for non‑trained users, in line with standards such as ISO 11608 and IEC 62366-1.
RD8 supports auto‑injector development through mechanical design, risk‑based decision‑making, and verification of functional performance and usability, helping teams establish confidence in device solutions progressing towards clinical, regulatory, and industrial implementation.

Prefilled Syringe Devices
Prefilled syringe devices are primary drug delivery systems designed to deliver a fixed, accurate dose via manual injection or integration into secondary delivery devices. They play a central role in injection‑based therapies by combining the drug container and delivery interface in a ready‑to‑use format that supports dose accuracy and simplified handling.
Engineering challenges for prefilled syringes centre on container–closure interactions, tolerance integration, and control of break‑loose and glide forces. Consistent performance across shelf life and use conditions is critical, alongside compliance with standards such as ISO 11040 and applicable parts of ISO 11608.
RD8 supports development through system‑level design activities, performance evaluation, and risk‑based verification, helping teams address integration constraints and ensure functional robustness as solutions advance towards clinical, regulatory, and industrial implementation.

Wearable Injectors
Wearable injectors are drug delivery devices designed to deliver precise doses over extended periods while attached to the patient’s body. They enable administration of larger volumes or controlled infusion profiles that cannot be achieved with conventional manual injections, supporting increasingly patient‑centric injection‑based therapies.
These systems combine fluid delivery with on‑body fixation, user interfaces, and often electromechanical control. Development challenges include controlled flow generation, adhesion performance, power management, and maintaining reliable operation under movement and environmental variability, alongside compliance with ISO 11608 and IEC 62366.
RD8 supports wearable injector development through system‑level design, mechanical and electromechanical integration, and verification of functional performance and usability, helping teams manage complexity and bring robust on‑body solutions towards implementation.

Custom Injection-Based Drug Delivery Systems
Custom injection‑based drug delivery systems are purpose‑built device solutions developed to address therapeutic, technical, or use‑context requirements that cannot be met by established delivery platforms. They support precise dose delivery in injection‑based therapies where differentiation or novel administration approaches are required.
Such systems are characterised by bespoke mechanical and system architectures tailored to specific dosing profiles, actuation principles, or integration constraints. Development is typically challenged by parallel technical uncertainties, evolving requirements, and the need to balance innovation with regulatory and manufacturability considerations.
RD8 supports custom system development through concept definition, system architecture development, and structured design and verification activities, enabling teams to navigate uncertainty and advance tailored device solutions towards clinical, regulatory, and industrial readiness.
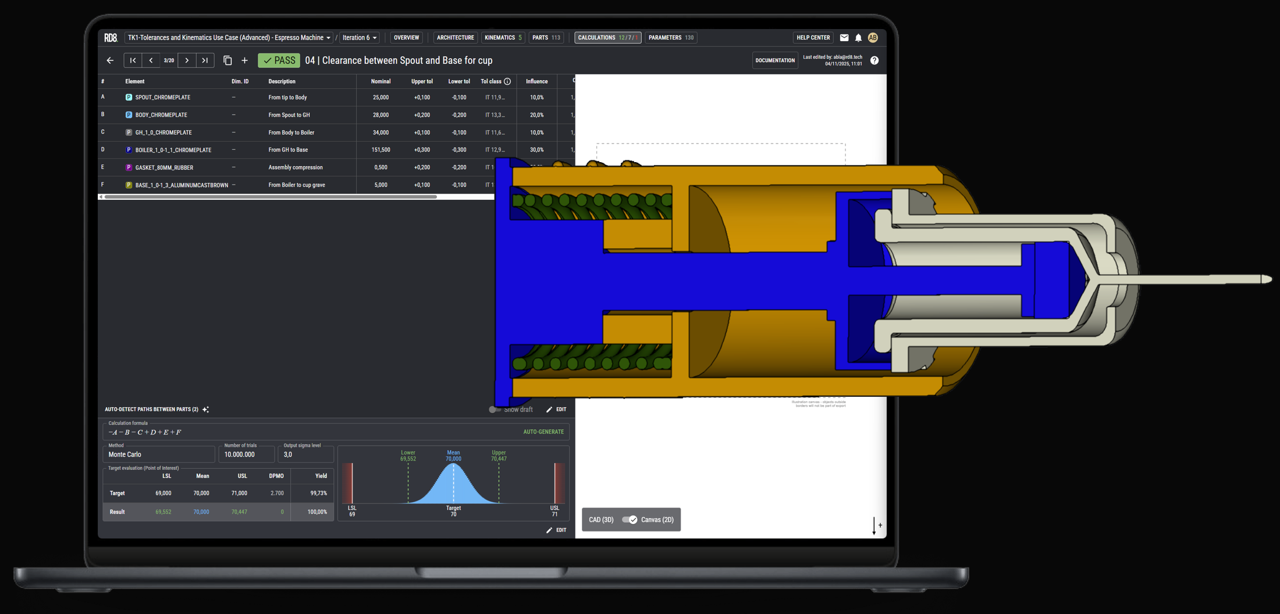
What Engineering Approach Does RD8 Use for Drug Delivery Device Development?
RD8 applies a systematic, data‑driven engineering approach to drug delivery device development, grounded in robust design principles and clear architectural thinking.
We are data driven, systematic and objective in our design processes, rooted in Robust Design principles and interface-based design optimisation and risk management
How Does Mechanical Engineering Support Each Stage of Drug Delivery Device Development?
Drug delivery device development progresses through defined stages, where decisions made early in the process directly influence downstream performance, risk, and manufacturability. Mechanical engineering provides the technical structure that links user needs and system requirements to detailed design, verification, and production‑ready solutions.
RD8 applies mechanical engineering in a stage‑appropriate manner, ensuring that each development phase builds logically on the previous one, reduces uncertainty, and enables controlled progression towards clinically and industrially robust device outcomes
1. Concept and Architecture Development
2. Prototype Development
3. Design Maturation
4. Product Readiness
We work with the best companies






What is mechanical engineering consulting
for drug delivery devices?
Whether you are developing a new device, a platform or upgrading a legacy design, drug delivery devices can be a challenge.
Engineering for drug delivery devices requires more depth and control than general mechanical engineering. All the way from requirement specification to verification and validation.
Mechanical engineering in this field requires high-performance solutions, designs that can scale to massive production volumes all the while maintaining traceability on design decisions and ensuring reliability.
To succeed you need System Engineering driven into functional architecture, a design process that supports risk management and a systematic, data driven approach to solution generation and maturing of your design.
RD8's Engineering Ecosystem [link] and design framework support development teams and enables development speed and inherent quality by design.
We work with you from early stage to industrialisation and ramp-up to ensure your device gets through approval and to market.
We deliver with your team to solve industry challenges like emergency device reliability levels, ultra-high production volumes or clearly defined risk-based design process
Architecture, Tolerances, and Robustness
R&D teams need clear product architecture based on first principles and clear specifications, rather than floating system requirements difficult to place and verify.
Building a solid architectural foundation allows doe tighter control of interactions between components by reducing unclear geometry and intended interfaces and interactions that may occur due to tolerances and variation.
This approach ensures robustness of interfaces is analysed, designed and verified up front rather than buried deep in high-level system interactions
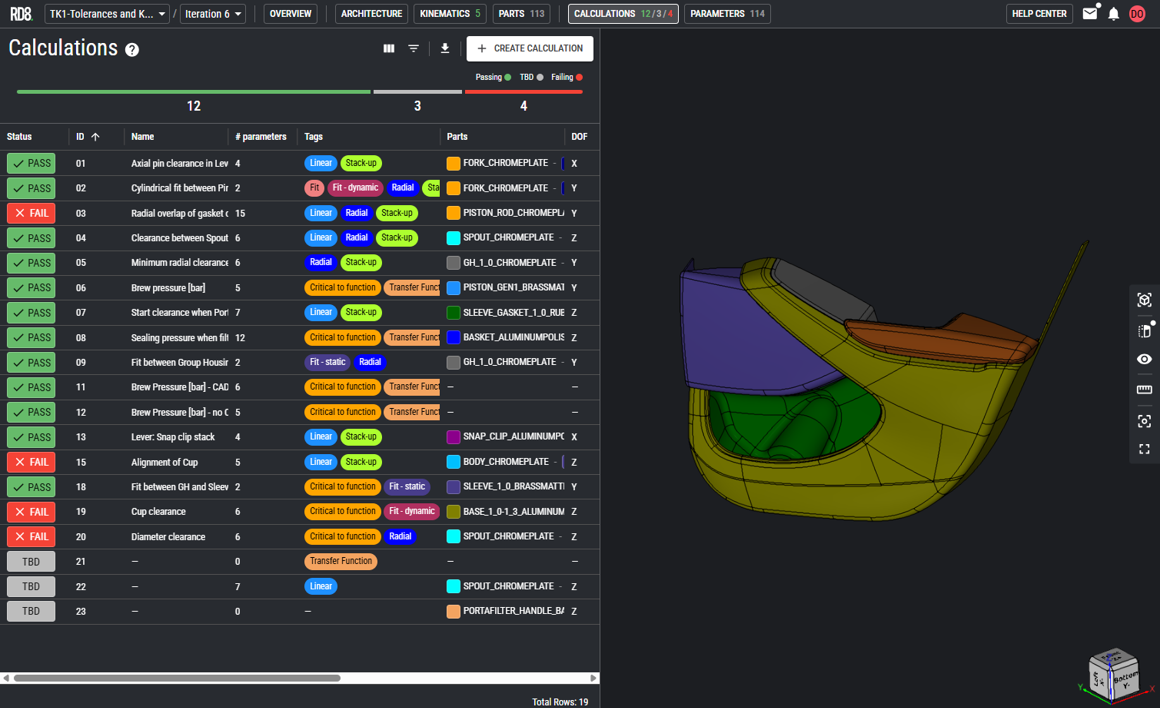
Concept-to-Production Support
We offer services from early stage concept development to industrialisation and ramp-up. Either working side-by-side in co-development or delivering work packages where you need it.
Sometimes, risk transparency and momentum can be created through a Design Assessment driven off our Engineering Ecosystem and metrics.
Our SMEs are here to help you tackle your challenges in device design

What mechanical engineering services are provided for drug delivery devices?
Device Architecture Development
Mechanical System Architecture
Functional Allocation Across Device Components
Component and Interface Definition
Mechanism Engineering
Injection and Activation Mechanism Design
Force, Alignment, and Dose Output Control
Tolerance and Variation Management
Dimensional Stack-Up Analysis
Injection Molding Variation Assessment
Device Performance Sensitivity Analysis
Robustness and Reliability Engineering
Engineering Margin Analysis
Robustness Across Use and Manufacturing Conditions
Reliability Improvement Actions
Failure Mode and Root-Cause Analysis
Failure Mode Identification
Root-Cause Investigation
Design Mitigation Strategies
Design for Manufacturability
Design for Injection-Molded Assemblies
Assembly Simplification
Production Tolerance Alignment
What Outcomes Does Mechanical Engineering Consulting Help Achieve for Drug Delivery Devices?
What mechanical engineering expertise does rd8 bring to drug delivery device development?
RD8 applies advanced mechanical engineering expertise to drug delivery devices, enabling reliable and predictable performance, controlled risk, and scalable manufacturability through systematic, data‑driven development
Device and Mechanism Architecture Expertise
This expertise enables clear functional decomposition, controlled interfaces, and early identification of technical constraints and risks.
A robust architecture is critical in drug delivery devices, as early structural decisions directly determine performance potential and stability, design flexibility, and the ability to manage complexity throughout development

Tolerance, Sensitivity, and Root-Cause Analysis Expertise
This expertise enables targeted design decisions based on quantitative understanding of sensitivities rather than trial‑and‑error iteration.
In drug delivery devices, where small variations can impact dose accuracy, activation force or other usability, this capability is essential for achieving predictable performance and avoiding late‑stage issues

Injection-Molded Assembly and Interface Design Expertise
This enables reliable part interaction, stable assembly behaviour, and alignment between design intent and manufacturing capability.
Interface quality is particularly critical in high‑volume drug delivery devices, where small inconsistencies at part boundaries can drive functional variation, assembly challenges, or reliability issues at scale - usually very difficult to troubleshoot

Robustness and Reliability Engineering Expertise
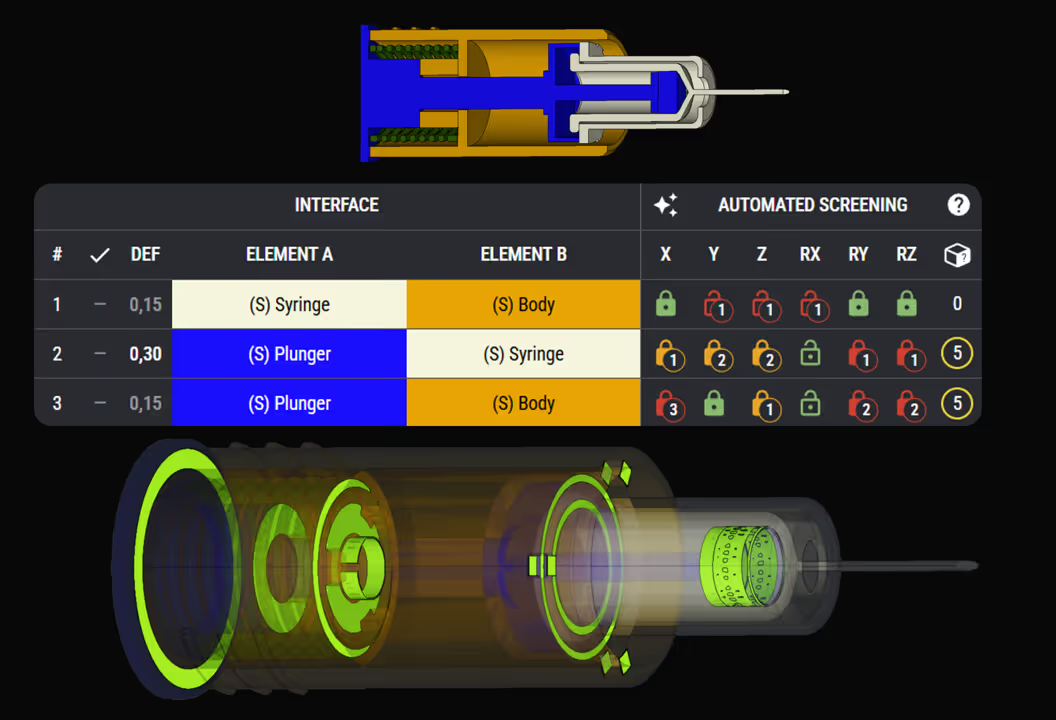
Who works with RD8 for mechanical engineering consulting?
RD8 works with organisations facing complex drug delivery device challenges where high reliability, controlled risk, and scalable production are critical to development success.
When application or timeline is must-not-fail and calls for advanced engineering support
Pharmaceutical R&D Teams
Drug Delivery Device Manufacurers
Mechanical engineering consulting supports optimisation of mechanisms, interfaces, and tolerances to ensure consistent performance and manufacturability.
RD8 can engage with both R&D and Manufacturing depending on need
Combination Product Development Teams
Medical Device and MedTech Organizations
How do teams work with rd8?
Why do teams work with rd8?
How can you start a drug delivery device engineering project?
1. Architecture and Design Assessment

- 1-8 engineering disciplines in assessment, including workshop sessions, handover and read-out, executive summary and detailed reporting- 3-6 weeks delivery time depending on scope
What you get
- Objective benchmark of your product’s design quality
- Visibility into hidden risks and overconstraints
- Data-backed recommendations for improved robustness and risk profile
- A trackable KPI to guide future development
2. Co-Development Support

- Define project objectives and
outcomes
- Mobilise, onboard and embed an expert team to your development
- Work based on client development model and governance (QMS, documentation standards, etc.)
What you get
- Transparent, data driven development support and deliverables
- Risk-based design and documentation progression
- Designs ready to scale with your manufacturing system
3. Long-Term Engineering Partnership

- Define strategic objectives and outcomes
- Tailor partner support across full RD8 portfolio (Engineering services, Academy, Software tools)
- Execute support and implementation programme with governance and portfolio metrics
What you get
- Design and risk transparency across portfolio
- Up-skilling of key design engineering competences
- integration to your development model, risk management and documentation practice
- Capacity smoothing and expert support on your pipeline
Frequently asked questions about drug delivery device engineering and development consulting
Below you will find frequently asked questions about Drug Delivery Devices and Development Consulting. If you have any further questions about how RD8 can assist in your design proces, please reach out to us.
How Does Mechanical Engineering Improve Drug Delivery Device Performance?
Mechanical engineering improves drug delivery device performance by designing and optimising device architecture, mechanisms, and interfaces so critical functions—such as dose delivery, actuation, and user interaction—remain predictable under real‑world variation. In drug delivery device development, tolerance engineering, sensitivity analysis, and verification‑driven design reduce performance drift caused by manufacturing variation, friction, wear, and environmental conditions, enabling reliable, production‑ready device performance at scale
What Are the Most Common Mechanical Engineering Challenges in Drug Delivery Devices?
This is highly device and application dependent. However, the most common challenges in drug delivery device mechanical design include controlling tolerances and variation, achieving stable force transmission and alignment, preventing interface‑driven functional sensitivity, and ensuring consistent mechanism behaviour over the device lifecycle. These challenges increase in high‑volume injection devices where tooling variation, assembly process variation, and long‑term reliability targets require robust design and design for manufacturability (DFM) to avoid late‑stage redesign and production instability
Does RD8 Provide Training or Academy Programs for Engineering Teams?
Yes. RD8 offers engineering training and academy programmes focused on robust design disciplines, including topics such as kinematics, tolerance design, DFM/DFA, and functions & sensitivity, with both on‑site and virtual learning formats available.
These programmes are designed to build in‑house engineering capability and are applied across industries including MedTech and drug delivery device development, with courses ranging from introductory to advanced practitioner levels
Does RD8 Offer Tolerance Stack-Up Analysis Software?
Yes. RD8 provides proprietary tolerance and interface analysis software that operates directly on 3D CAD data to identify interfaces, stack‑ups, sensitivities, and improvement potential. The software supports automated tolerance calculations, variation analysis, and live collaboration, enabling teams to improve design quality and reduce trial‑and‑error during development
Get in touch with our experts
Let's share viewpoints on next level design quality.
Shoot us a message and we will get back as soon as possible.
By submitting, you accept RD8's Privacy Policy and Terms of Service.


